Covid Corner News
Featured Article

Featured Article

Providing a sense of hope and relief during the pandemic
The development of the first safe and effective coronavirus disease 2019 (COVID-19) vaccine has been the most anticipated discovery in recent health care history. The pandemic also brought about a unique opportunity for pharmacists to step up and work to protect public health, as the vaccines received Emergency Use Authorization from the Food and Drug Administration. Read the ArticleFeatured Article
Pfizer-BioNTech Vaccine for COVID-19 Gains Approval for Use in Patients Aged > 12 Years of Age
Author: Kyra Browne, PharmD – PGY-1 Resident Howard University Hospital Last week, the Pfizer-BioNTech vaccine for the prevention of COVID-19 became the first out of the three currently available FDA-authorized vaccines in the U.S. to receive Emergency-Use Authorization in patients aged 12 to 15 years of age. Initial EUA recommendations were made for patients aged 18 and older, which was extended to patients aged 16 and older in December. Read the ArticleFeatured Article

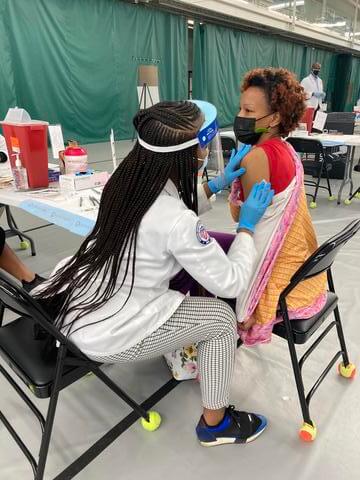
Students Providing Covid Testing and Vaccinations at Grubb's Care Pharmacy and Morgan's Pharmacy
Dr. Unonu with P4 student Patrick Fotso at Grubb's Care Pharmacy Ambulatory Care rotation site in NE DC. At this location, they are providing COVID-19 antibody, 24-hour turnaround PCR, and 20-minute turnaround rapid antigen testing to the community in addition to COVID-19 vaccinations. The tests and vaccinations are provided every 5 minutes and the patients have abundantly expressed their gratitude for providing this service to the community. Read the ArticleFeatured Article
Just the Facts: Johnson & Johnson COVID-19 Vaccine
Causing undue harm to any patient goes against the code of every health care worker. While we often have to weigh the risks vs. benefits of different procedures and medications, we are also responsible for making sure we have the most up-to-date information for our patients. It is our professional duty to uphold the standards of care based on scientific evidence and reason. On April 13, 2021, the CDC and FDA recommended a pause in the use of Johnson’s & Johnson’s Janssen COVID-19 Vaccine (J&J Vaccine). Read the ArticleFeatured Article